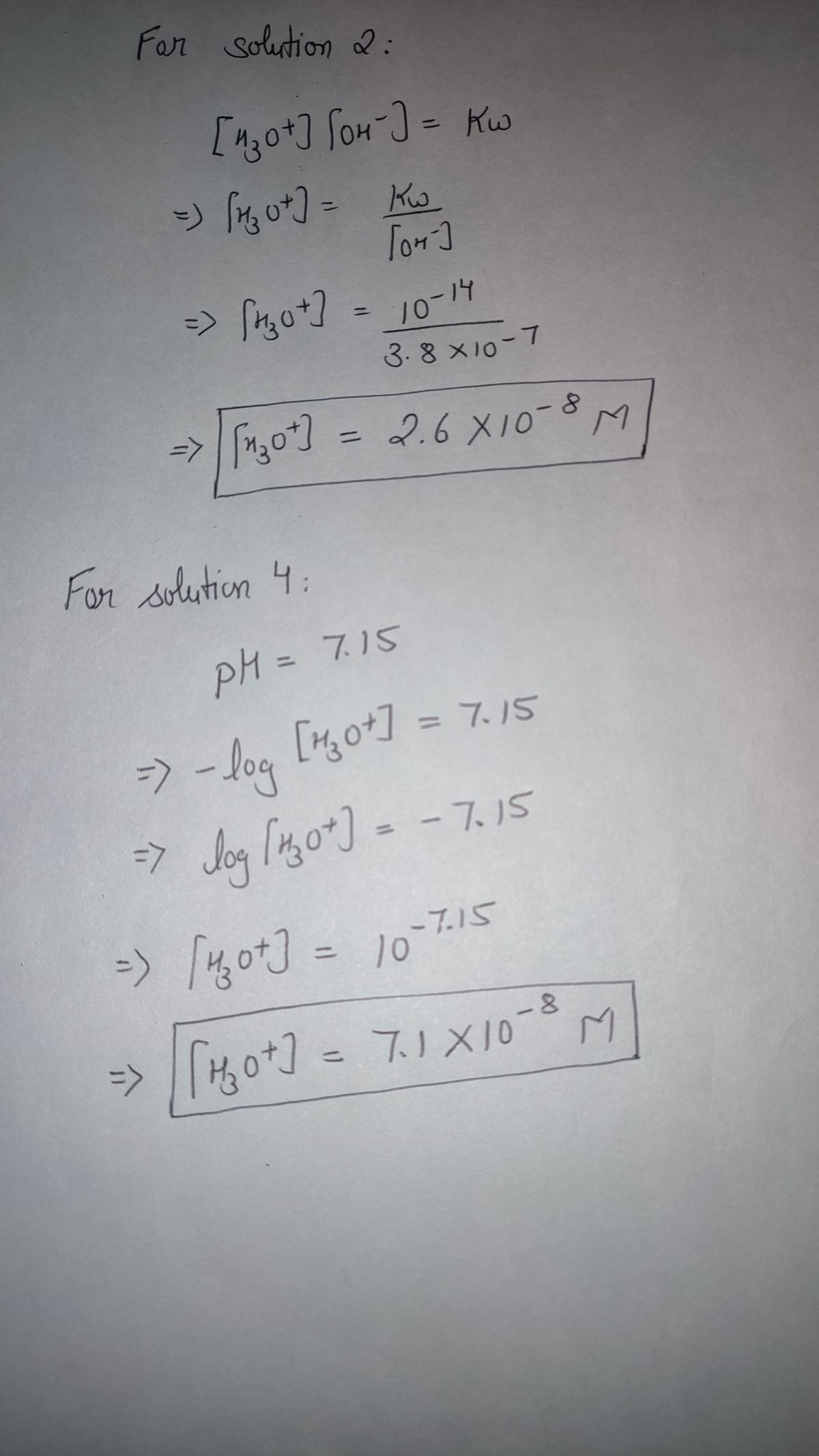31 0.1 ml 0.001 M hcl solution is diluted with water to make 10 litres .calculate PH of the dilute solution.

Assertion : `pH` of `10^(-7) M HCl` is less than `7` at `25^(@)C`. Reason : At very low concentratio - YouTube
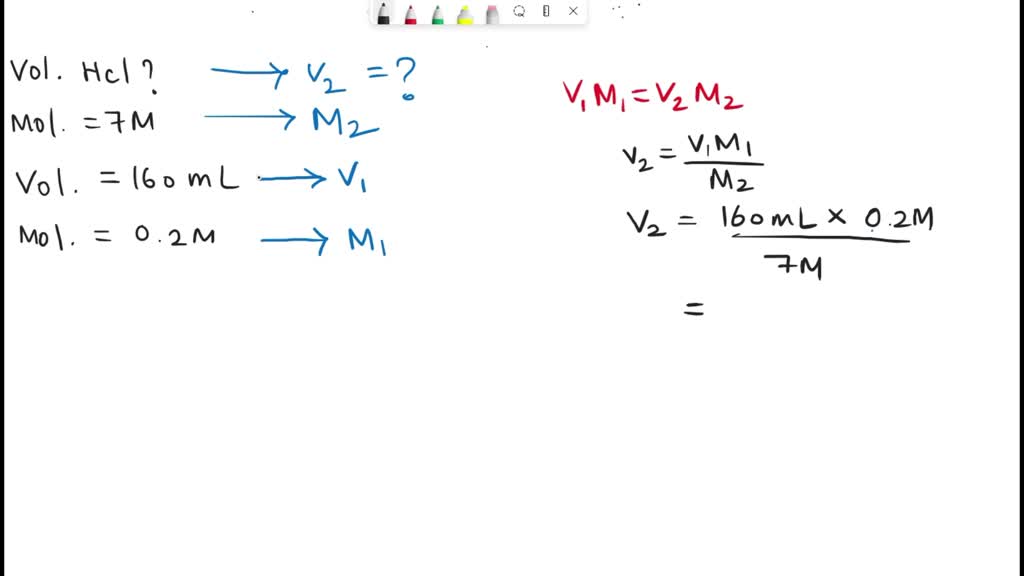
SOLVED: 2. How many ml of 7 M HCL solution is required to prepare 150 mL of 0.2 M HCL solution? Calculate the pH of the resulting dilute acid solution.

10^{-5}space Mspace NaOH solution 25^{smallcirc}C is diluted 1000space times. The pH of the resultant solution will:be equal to 8lie between 7 and 8lie between 6 and 7remain unchanged
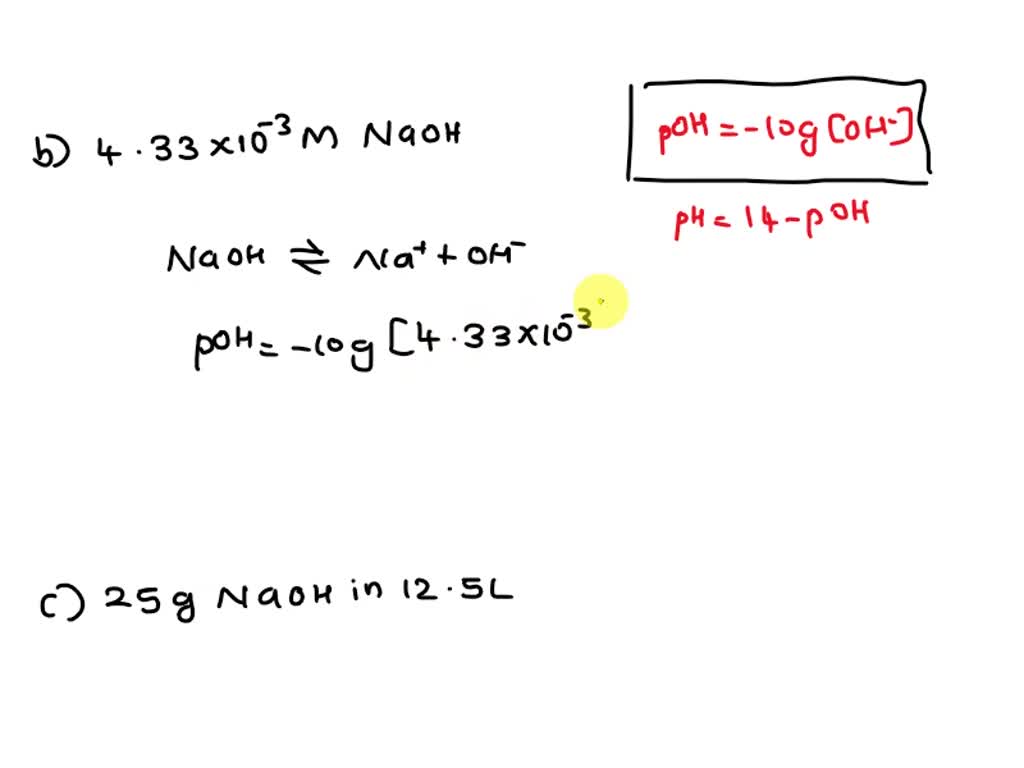
SOLVED: 7) What are the pH values of the following solutions? a) A 1.45 x 10-5 M HCl solution. b) A 4.33 x 10-3 M NaOH solution. c) A solution that contains


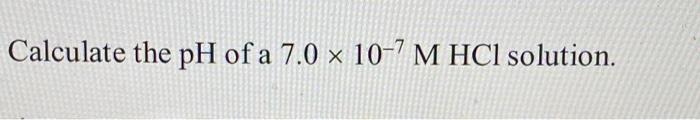



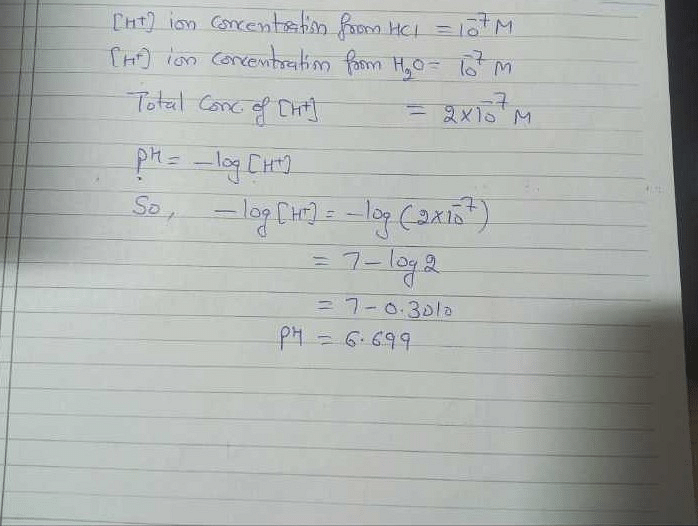
![Malayalam] The pH of a 10^(-7) M aq. solution of HCl at 298 K is 7 Malayalam] The pH of a 10^(-7) M aq. solution of HCl at 298 K is 7](https://static.doubtnut.com/ss/web/9899943.webp)
![Bengali] What is the pH of 10^(-7)(M) HCl solution- Bengali] What is the pH of 10^(-7)(M) HCl solution-](https://static.doubtnut.com/ss/web/3771129.webp)
:max_bytes(150000):strip_icc()/how-to-calculate-ph-quick-review-606089_final-165915b0177b4f6e82843f25097f51df.png)

![Solved Calculate the pH of each solution. a) [H30+] = 1.7 x | Chegg.com Solved Calculate the pH of each solution. a) [H30+] = 1.7 x | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2F0fa%2F0fa98cc3-4673-4562-9448-02d65a91573c%2Fimage)