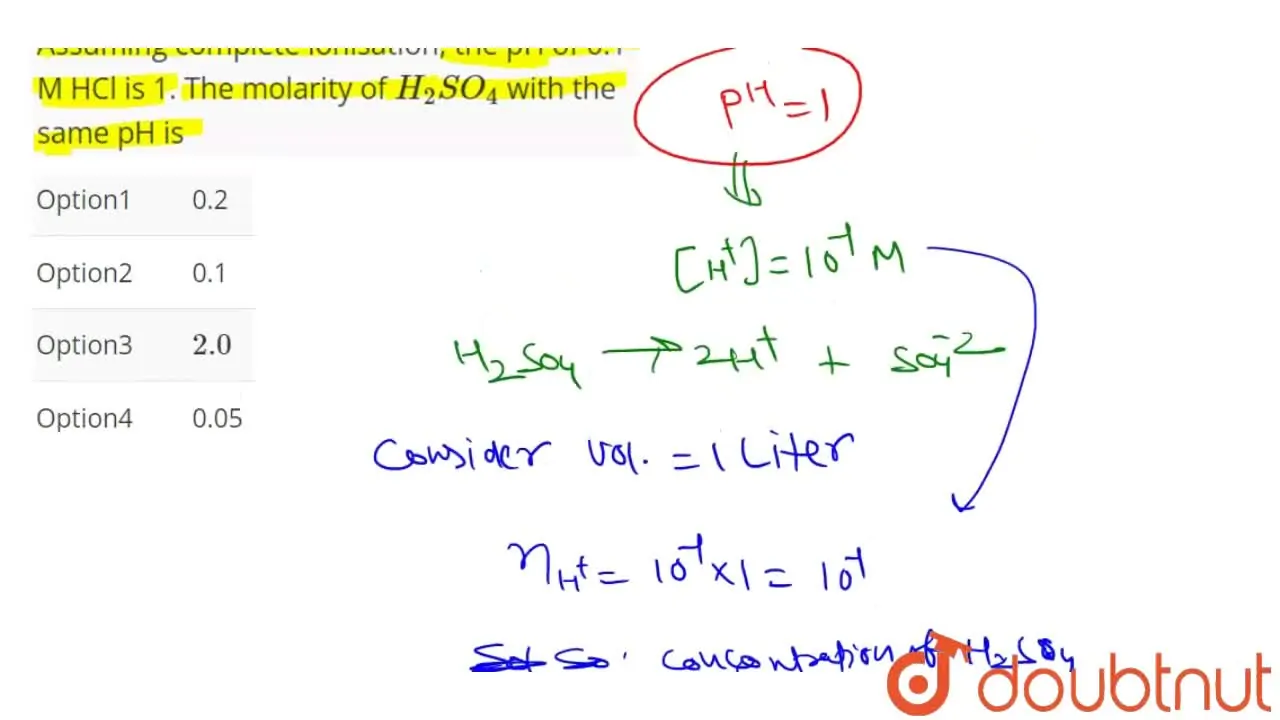
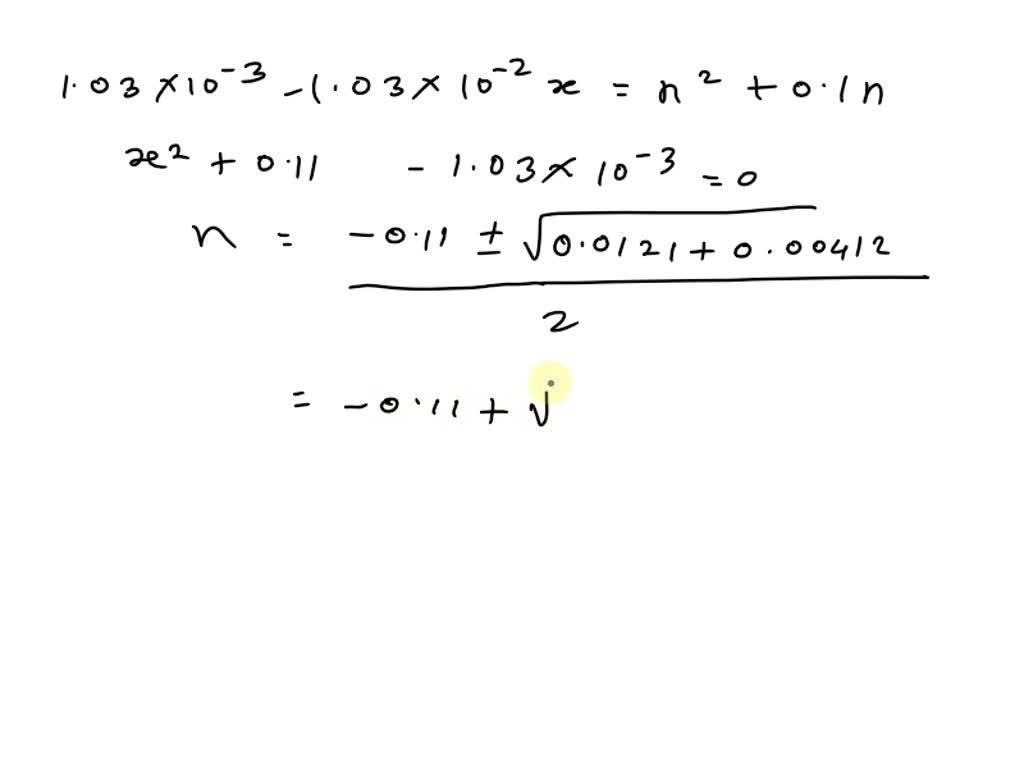Consider the following statements (a) The pH of a mixture containing 400 mL of 0.1 M H2SO4 - Sarthaks eConnect | Largest Online Education Community

a) Current density–potential curves in a 0.1 M H2SO4 aqueous solution... | Download Scientific Diagram

200ml N/20 HNO3, 100ml N/20 H2SO4, 300ml M/30 HCL, 50ml 0.1M Ba(OH)2 and 250 ml M/10 NaCl solutions were mixed then value of pH of resultant solution is:- (1) 1.65. (2) 2.0 (3). 1.78. (4) 0.16? - EduRev NEET Question
Calculate pH of following solutions:0.1 M { H }_{ 2 }S{ O }_{ 4 }left( 50 ml right) +0.4 M HCl 50left( ml right) quad left[ log { 0.3=-0.522 } right]
![SOLVED: Calculate the pH of a 0.1 wt% H2SO4 (sulfuric acid) solution. You can assume that the density of the solution is that of water. Must use pH = -log[H+]. Please explain SOLVED: Calculate the pH of a 0.1 wt% H2SO4 (sulfuric acid) solution. You can assume that the density of the solution is that of water. Must use pH = -log[H+]. Please explain](https://cdn.numerade.com/ask_previews/fb910f96-3dc9-421e-9ac5-8d5a0aaf4d70_large.jpg)
SOLVED: Calculate the pH of a 0.1 wt% H2SO4 (sulfuric acid) solution. You can assume that the density of the solution is that of water. Must use pH = -log[H+]. Please explain

10 ml of 0.1 M H2SO4 is mixed with 20 ml of 0.1 MKOH, the pH of resulting solution will be(a) 0(b) 7(c) 2(d) - Brainly.in

600 mL of 0.01 M HCl is mixed with 400 mL of 0.01 M H2SO4. The pH of the mixture is _____ × 10−2. - YouTube